SUPERIOR EFFICACY ACROSS LINES OF THERAPY
Sustained efficacy at ~6-year milestone in 1L and ~3.5-year milestone in 2L
1L: SEQUOIA
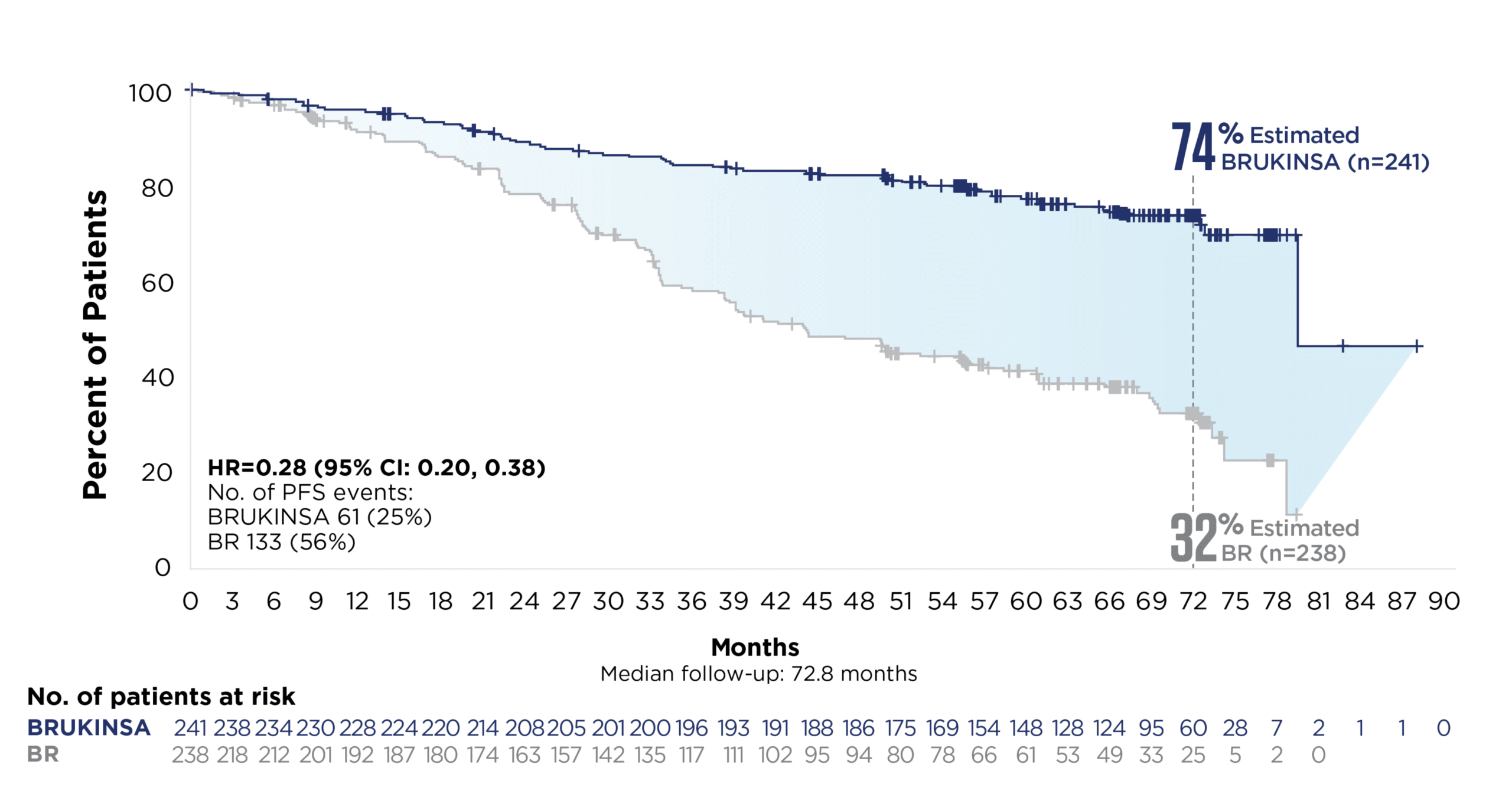
SUPERIOR PFS vs BR IN PATIENTS WITHOUT DEL(17p); SUSTAINED RESULTS AT THE ~6-YEAR MILESTONE1-3
58% relative risk reduction in disease progression or death with BRUKINSA vs BR at the superiority analysis
(Cohort 1; primary endpoint; HR=0.42; 95% CI: 0.28, 0.63; p<0.0001; median follow-up: 26.2 months)*†
72% relative risk reduction in disease progression or death with BRUKINSA vs BR at ~6 years‡
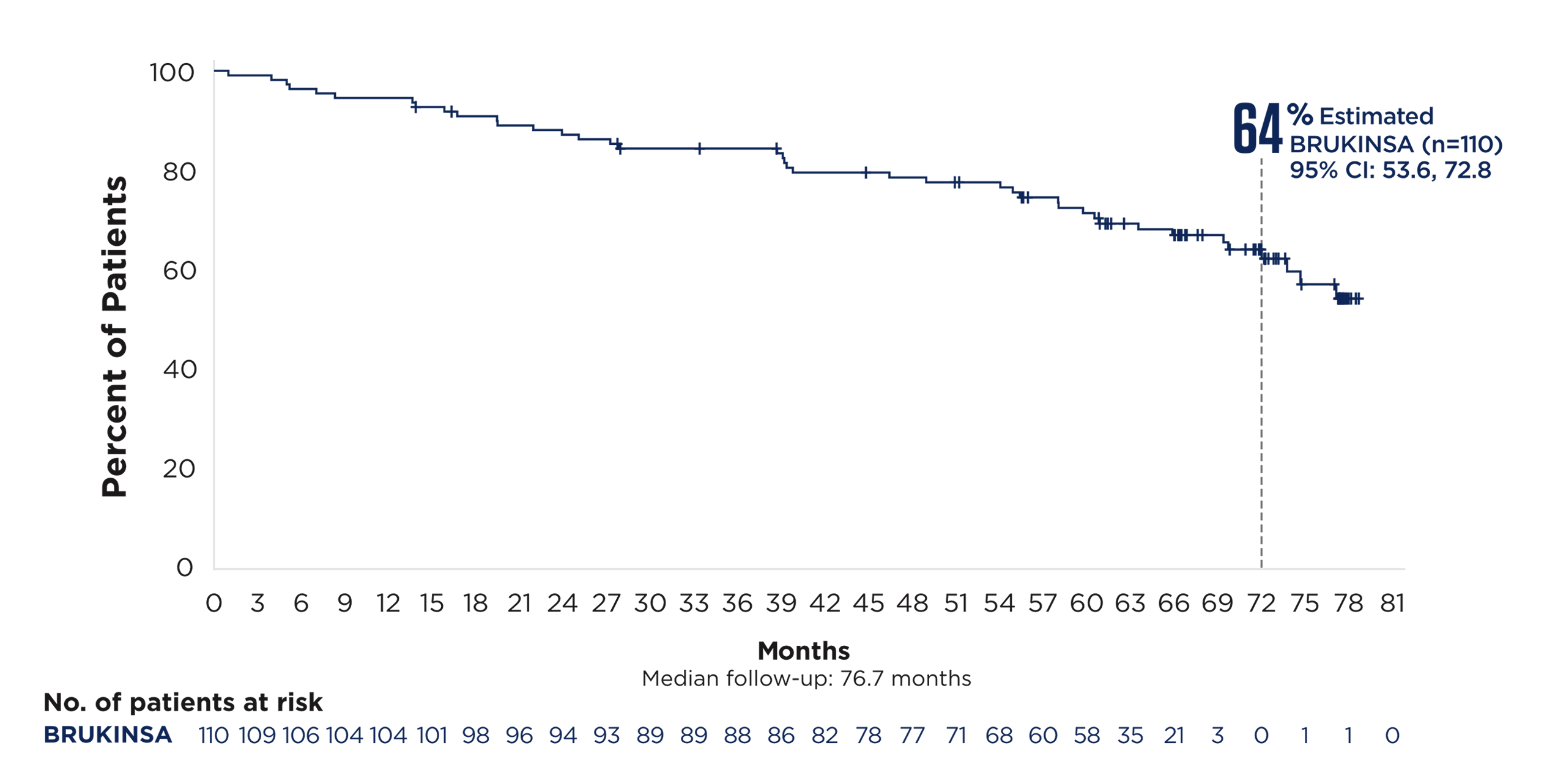
Cohort 1: patients without del(17p)
CONSISTENT PFS IN PATIENTS WITH DEL(17p) +/-TP53 AND ulgHV3-8
In the largest prospective study of 1L CLL patients with del(17p), BRUKINSA
delivered an unmatched PFS rate at ~6 years§3-8
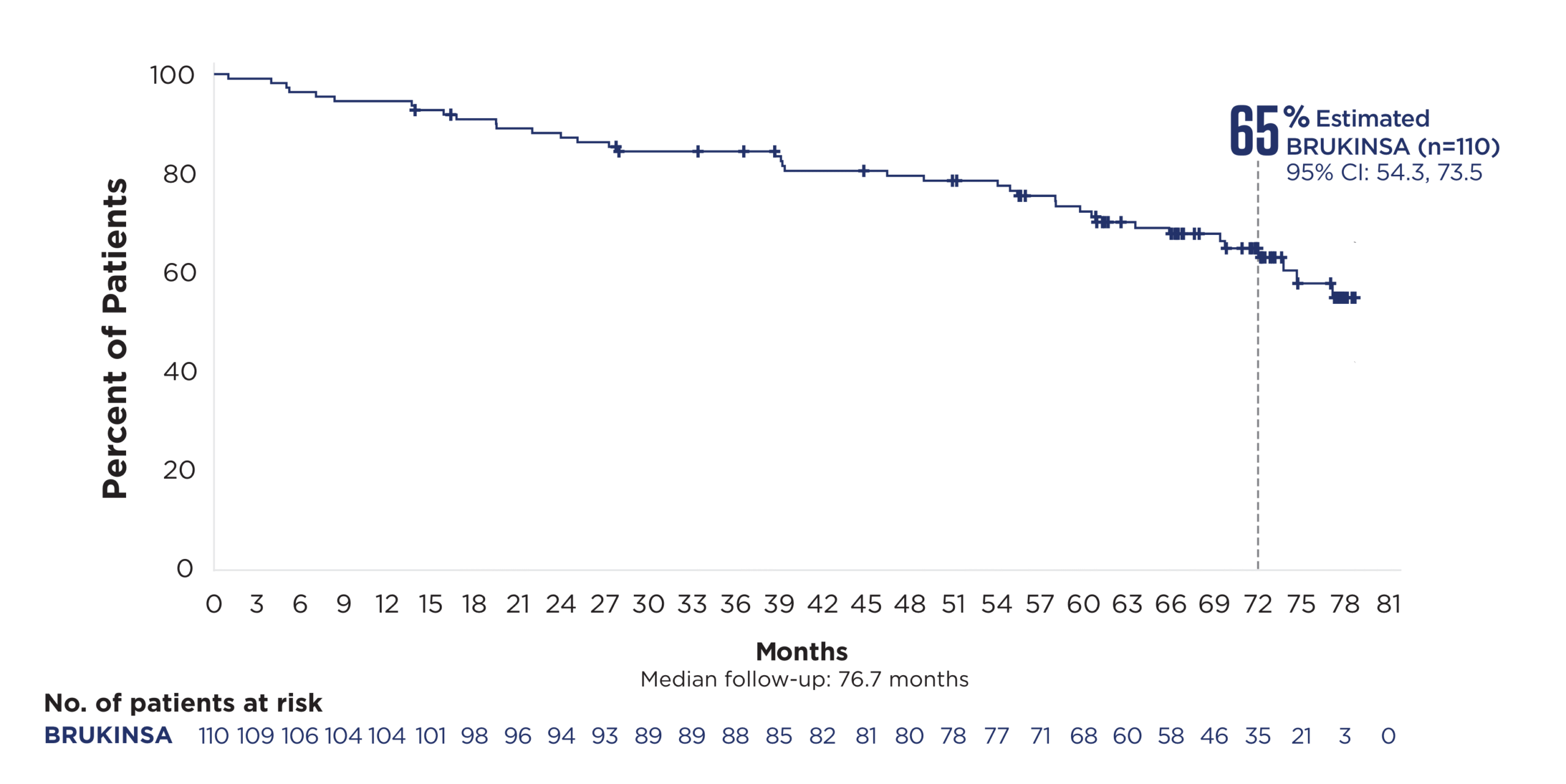
Cohort 2: patients with del(17p)
78% relative risk reduction in
disease progression or death with
BRUKINSA vs BR in patients with uIgHV at ~6 years§3
Cohort 1: patients without del(17p)
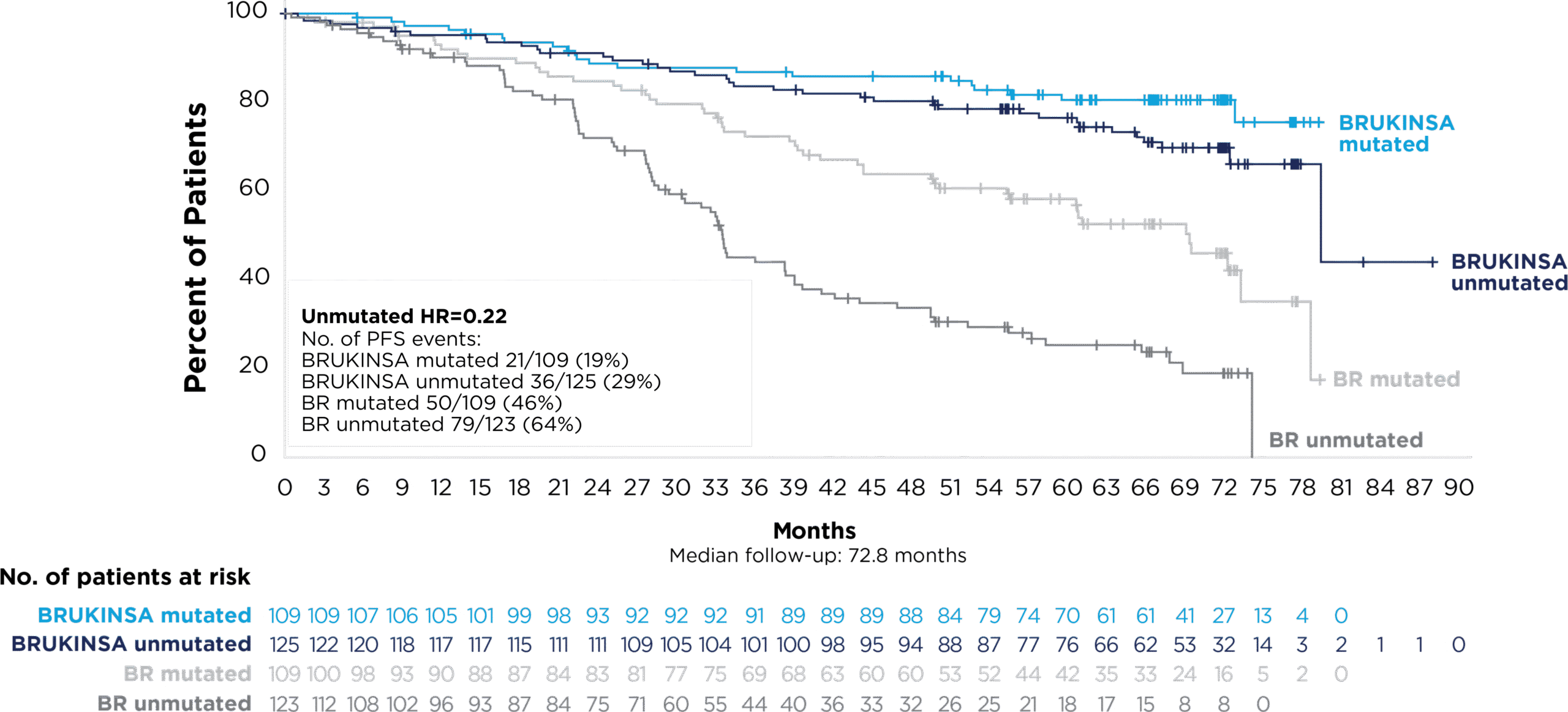
IN PATIENTS WITHOUT DEL(17p):
PFS GENERALLY FAVORED BRUKINSA AT ~5 YEARS, REGARDLESS OF MUTATION STATUS*†9
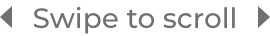

Complete responses deepened over time in patients without and with del(17p)1-3



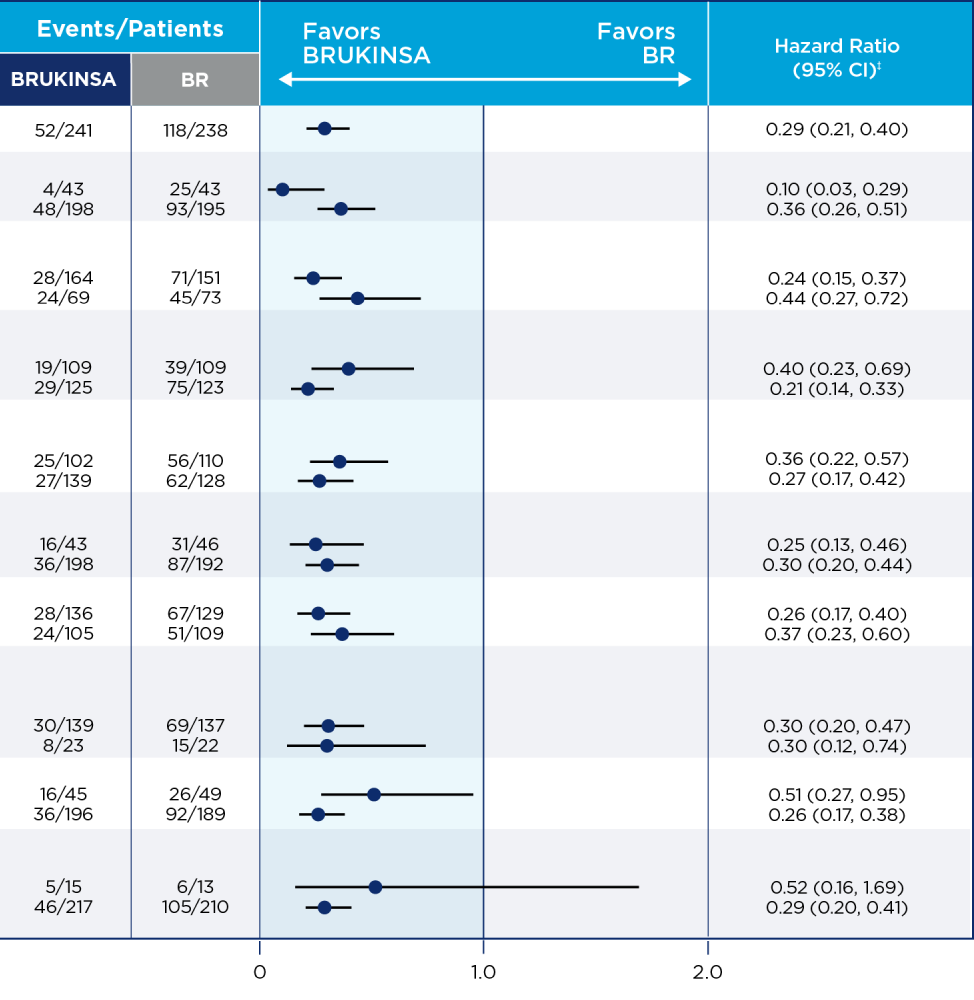
THE ONLY BTKi WITH SUPERIOR EFFICACY vs IBRUTINIB; SUSTAINED RESULTS AT THE ~3.5-YEAR MILESTONE1,10,11
35% relative risk reduction in disease progression or death with BRUKINSA vs ibrutinib in all-comers at the superiority analysis
(secondary endpoint; HR=0.65; 95% CI: 0.49, 0.86; p=0.0024; median follow-up: 31 months)*
32% relative risk reduction in disease progression or death with BRUKINSA vs ibrutinib at ~3.5 years†
all-comer population
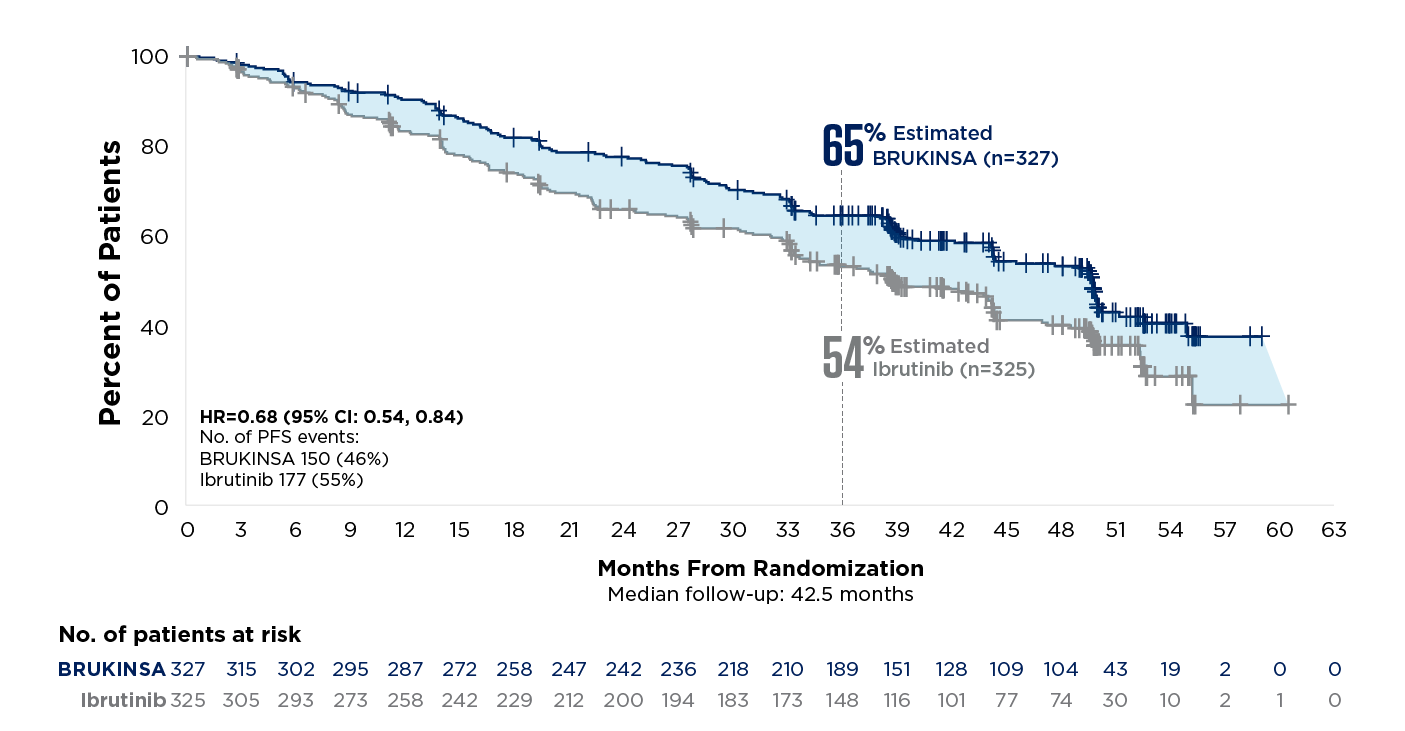
49% relative risk reduction in disease progression or death vs ibrutinib†
patients with del(17p) +/-TP53
CONSISTENT PFS vs IBRUTINIB AT INITIAL ANALYSIS, REGARDLESS OF MUTATION STATUS*†10
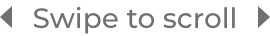

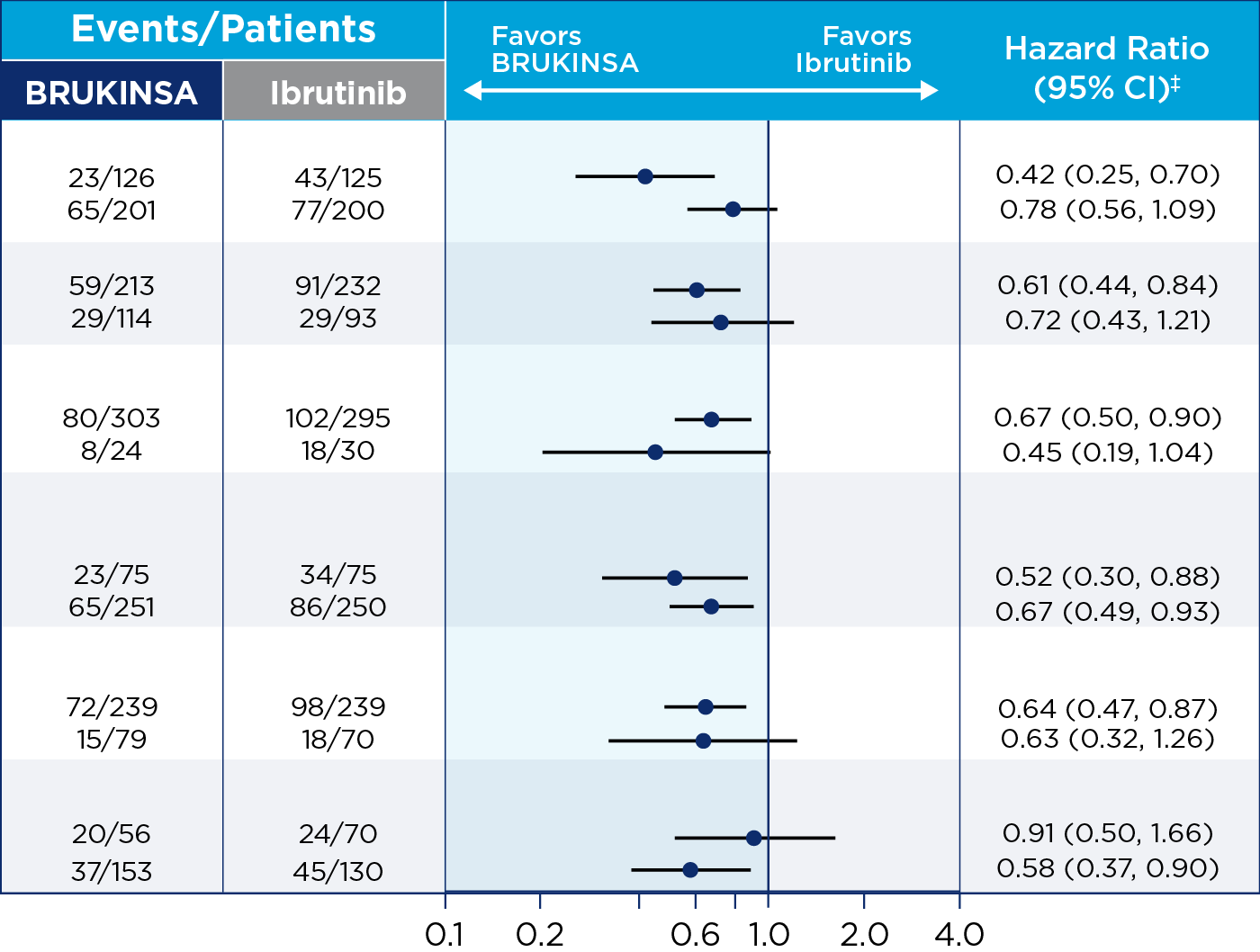
THE ONLY BTKi TO ACHIEVE SUPERIOR ORR vs IBRUTINIB1
Initial analysis (25 months for ORR)
BRUKINSA DEMONSTRATED MORE COMPLETE RESPONSES OVER TIME vs IBRUTINIB11

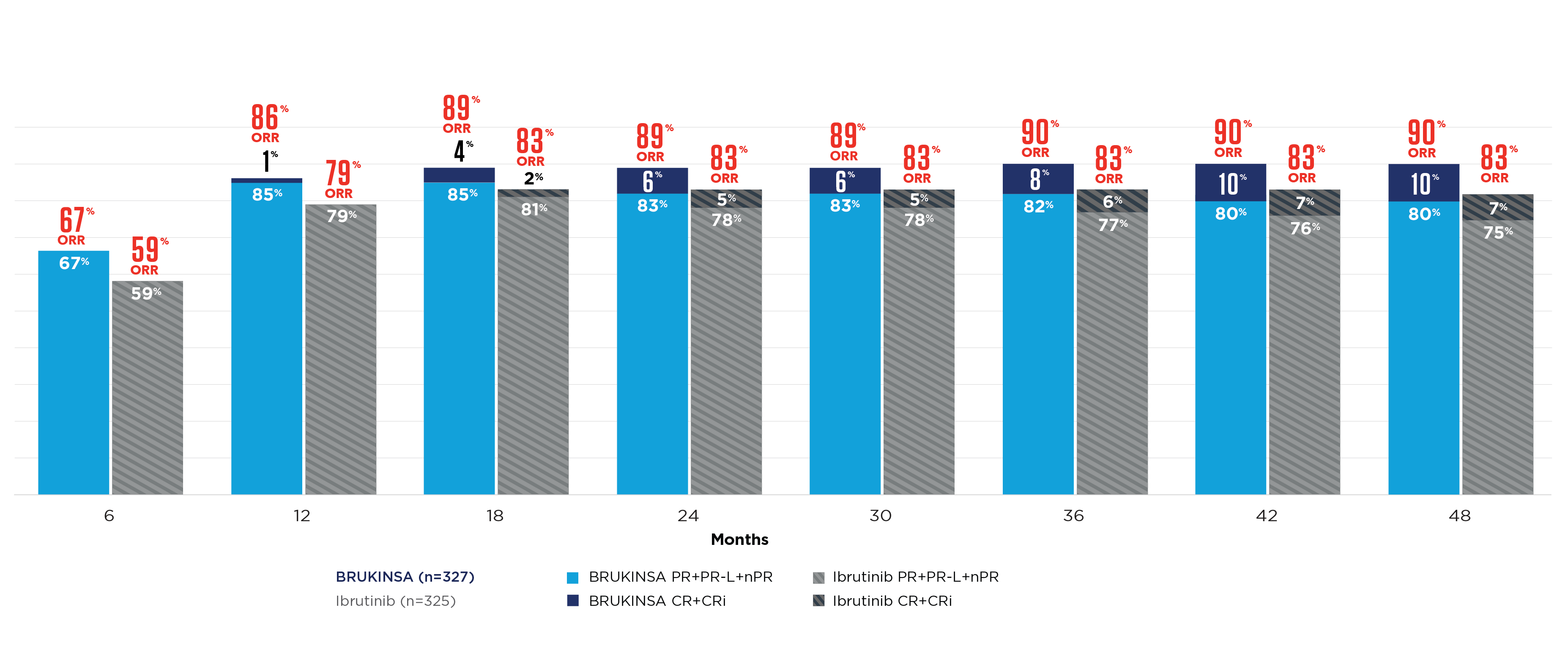
CONSISTENT ORR vs IBRUTINIB AT INITIAL ANALYSIS, REGARDLESS OF MUTATION STATUS10

Dr Anthony Nguyen discusses efficacy data across mutations in CLL
References: 1. BRUKINSA. Package insert. BeOne Medicines USA, Inc.; 2025. 2. Tam CS, Brown JR, Kahl BS, et al. Zanubrutinib versus bendamustine and rituximab in untreated chronic lymphocytic leukaemia and small lymphocytic lymphoma (SEQUOIA): a randomised, controlled, phase 3 trial. Lancet Oncol. 2022;23(8):1031-1043. 3. Tam CS, Munir T, Robak T, et al. Sustained efficacy of zanubrutinib vs bendamustine + rituximab in treatment-naive chronic lymphocytic leukemia/small lymphocytic lymphoma and continued favorable survival in non-randomized patients with del(17p): 6-year follow-up in the phase 3 SEQUOIA study. Presented at: American Society of Hematology (ASH) Annual Meeting and Exposition; December 6-9, 2025. Poster 2129. 4. Shadman M, Munir T, Ma S, et al. Combination of zanubrutinib + venetoclax for treatment-naive CLL/SLL: results in SEQUOIA Arm D. Presented at: American Society of Clinical Oncology (ASCO) 2025 Annual Meeting; May 30-June 3, 2025. 5. Davids MS, Ryan CE, Lampson BL, et al. Phase II study of acalabrutinib, venetoclax, and obinutuzumab in a treatment-naïve chronic lymphocytic leukemia population enriched for high-risk disease. J Clin Oncol. 2025;43(7):788-799. 6. Sharman JP, Egyed M, Jurczak W, et al. Acalabrutinib-obinutuzumab improves survival vs chemoimmunotherapy in treatment-naive CLL in the 6-year follow-up of ELEVATE-TN. Blood. 2025;146(11):1276-1285. 7. Al-Sawaf O, Robrecht S, Zhang C, et al. Venetoclax-obinutuzumab for previously untreated chronic lymphocytic leukemia: 6-year results of the randomized phase 3 CLL14 study. Blood. 2024;144(18):1924-1935. 8. Wierda WG, Jacobs R, Barr PM, et al. Outcomes in high-risk subgroups after fixed-duration ibrutinib + venetoclax for chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL): up to 5.5 years of follow-up in the phase 2 CAPTIVATE study. J Clin Oncol. 2024;42(16[suppl]). Abstract 7009. 9. Shadman M, Munir T, Robak T, et al. Zanubrutinib versus bendamustine and rituximab in patients with treatment-naïve chronic lymphocytic leukemia/small lymphocytic lymphoma: median 5-year follow-up of SEQUOIA. J Clin Oncol. 2025;43(7):780-787. 10. Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib or ibrutinib in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2023;388(4):319-332. 11. Brown JR, Eichhorst B, Lamanna N, et al. Sustained benefit of zanubrutinib vs ibrutinib in patients with R/R CLL/SLL: final comparative analysis of ALPINE. Blood. 2024;144(26):2706-2717. 12. Data on file. BeiGene USA, Inc.