DEMONSTRATED SAFETY ACROSS LINES OF THERAPY,
INCLUDING LOW RATES OF CARDIAC EVENTS IN 1L AND 2L CLL
1L: SEQUOIA
SEQUOIA (STUDY 304)
COHORT 1: OVERALL INCIDENCE OF ADVERSE REACTIONS (ARs)*1,2
| Adverse Reactions | ARs in ≥10% of Patients Without Del(17p) at the Initial Analysis |
Pooled Safety Population in All-Comers† |
||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=240) | BR (n=227) | BRUKINSA (N=1729) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Musculoskeletal pain | 33 | 2 | 17 | 0.4 | 24 | 2 |
| Upper respiratory tract infection | 28 | 1 | 15 | 0.9 | 38 | 3 |
| Pneumonia | 13‡ | 5 | 8§ | 4 | 17 | 11 |
| Hemorrhage | 27‡ | 4 | 4 | 0.4 | 32 | 4 |
| Hypertension | 14 | 7 | 5 | 3 | 14 | 7 |
| Rash | 24 | 1 | 30 | 5 | 25 | 0.6 |
| Bruising | 24 | 0 | 3 | 0 | 21 | 0.1 |
| Cough | 15 | 0 | 10 | 0 | 20 | 0.1 |
| Diarrhea | 14 | 0.8 | 12§ | 0.9 | 20 | 2 |
| Constipation | 10 | 0.4 | 18 | 0 | 13 | 0.3 |
| Nausea | 10 | 0 | 33 | 1 | 11 | 0.2 |
| Fatigue | 14 | 1 | 21 | 2 | 18 | 1 |
| Second primary malignancy | 13‡ | 6 | 1 | 0.4 | 15 | 7 |
| Headache | 12 | 0 | 8 | 0 | 11 | 0.3 |
| Dizziness | 11 | 0.8 | 5 | 0 | 11 | 0.3 |
SEQUOIA (STUDY 304)
COHORT 2: OVERALL INCIDENCE OF ARs*1,2
| Adverse Reactions | ARs in ≥10% of Patients With Del(17p) at the Initial Analysis |
Pooled Safety Population in All-Comers† |
||
|---|---|---|---|---|
| BRUKINSA (n=111) | BRUKINSA (N=1729) | |||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Upper respiratory tract infection | 38 | 0 | 38 | 3 |
| Pneumonia | 20‡ | 8 | 17 | 11 |
| Musculoskeletal pain | 38 | 3 | 24 | 2 |
| Rash | 28 | 0 | 25 | 0.6 |
| Bruising | 26 | 0.9 | 21 | 0.1 |
| Hemorrhage | 28 | 5 | 32 | 4 |
| Hypertension | 11 | 5 | 14 | 7 |
| Second primary malignancy | 22§ | 6 | 15 | 7 |
| Diarrhea | 18 | 0.9 | 20 | 2 |
| Nausea | 16 | 0 | 11 | 0.2 |
| Constipation | 15 | 0 | 13 | 0.3 |
| Abdominal pain | 12 | 2 | 11 | 0.9 |
| Cough | 18 | 0 | 20 | 0.1 |
| Dyspnea | 13 | 0 | 8 | 0.6 |
| Fatigue | 14 | 0.9 | 18 | 1 |
| Headache | 11 | 2 | 11 | 0.3 |
SEQUOIA (STUDY 304): INCIDENCE OF LABORATORY ABNORMALITIES*1
Select Lab Abnormalities (≥20%) That Worsened From Baseline in Cohorts 1 and 2 at the Initial Analysis
| Laboratory Abnormality | Cohort 1: Patients Without Del(17p) |
Cohort 2: Patients With Del(17p) |
||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=239)† | BR (n=227)† | BRUKINSA (n=111)‡ | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Neutrophils decreased | 37 | 15 | 80 | 53 | 42 | 19§ |
| Hemoglobin decreased | 29 | 3 | 66 | 8 | 26 | 4 |
| Platelets decreased | 27 | 2 | 61 | 11 | 23 | 0.9 |
| Leukocytes increased | 21¶ | 21 | 0.4 | 0.4 | NR | NR |
| Glucose increased# | 55 | 7 | 67 | 10 | 52 | 6 |
| Creatinine increased | 22 | 0.8 | 18 | 0.4 | 27 | 0.9 |
| Magnesium increased | 22 | 0 | 14 | 0.4 | 31 | 0 |
| Alanine aminotransferase increased | 21 | 2 | 23 | 2 | NR | NR |
SEQUOIA (STUDY 304): SELECT ADVERSE EVENTS (AEs) OF SPECIAL INTEREST*1-3
| Adverse Events | SEQUOIA (Initial Analysis) | Pooled Safety Population† | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=240) | BR (n=227) | BRUKINSA (N=1729) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Fatigue | 12 | 1 | 15 | 0.9 | 18 | 1 |
| Headache | 11 | 0 | 7 | 0 | 11 | 0.3 |
| Myalgia | 4 | 0 | 1 | 0 | 4 | 0.3 |
| Arthralgia | 13 | 0.8 | 9 | 0.4 | 14 | 0.6 |
| Atrial fibrillation | 3 | 0.4 | 3 | 1 | 4 | 2 |
| Hypertension | 14 | 6 | 11 | 5 | 14 | 7 |
| Major bleeding‡ | 5 | 4 | 2 | 2 | 5 | 4 |
SEQUOIA (STUDY 304):
LOWER RATES OF DOSE REDUCTIONS AND DISCONTINUATION DUE TO AEs AT THE INITIAL ANALYSIS3

SEQUOIA (STUDY 304):
LOW RATES OF AFIB IN PATIENTS WITHOUT DEL(17p) AT THE INITIAL ANALYSIS3

- Grade ≥3 rate of afib was 0.4% with BRUKINSA vs 1.3% with BR
ALPINE (STUDY 305): OVERALL INCIDENCE OF ADVERSE REACTIONS (ARs)*1,2
| Adverse Reactions | ARs in ≥10% of Patients at the Initial Analysis | Pooled Safety Population† | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=324) | Ibrutinib (n=324) | BRUKINSA (N=1729) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Upper respiratory tract infection | 27 | 1 | 22 | 1 | 38 | 3 |
| Pneumonia | 18‡ | 9 | 19§ | 11 | 17 | 11 |
| COVID-19 | 14‡ | 7 | 10§ | 5 | 10 | 5 |
| Musculoskeletal pain | 26 | 0.6 | 28 | 0.6 | 24 | 2 |
| Hemorrhage | 24‡ | 3 | 26§ | 4 | 32 | 4 |
| Hypertension | 19 | 13 | 20 | 13 | 14 | 7 |
| Rash | 20 | 1 | 21 | 0.9 | 25 | 0.6 |
| Bruising | 16 | 0 | 14 | 0 | 21 | 0.1 |
| Diarrhea | 14 | 2 | 22 | 0.9 | 20 | 2 |
| Fatigue | 13 | 0.9 | 14 | 0.9 | 18 | 1 |
| Cough | 11 | 0.3 | 11 | 0 | 20 | 0.1 |
| Dizziness | 10 | 0 | 7 | 0 | 11 | 0.3 |
Rates of hypertension were comparable between BRUKINSA and ibrutinib1
- A medical history of hypertension was reported in more than half of these patient events for both BRUKINSA and ibrutinib2
ALPINE (STUDY 305): INCIDENCE OF LAB ABNORMALITIES*1
Select Lab Abnormalities (≥20%) That Worsened From Baseline at the Initial Analysis
| Laboratory Abnormality | BRUKINSA (n=321) | Ibrutinib (n=321)† | ||
|---|---|---|---|---|
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Neutrophils decreased | 43 | 15 | 33 | 16 |
| Hemoglobin decreased | 28 | 4 | 32 | 4 |
| Lymphocytes increased | 24 | 19 | 26 | 19 |
| Platelets decreased | 22 | 4 | 24 | 3 |
| Glucose increased | 52 | 5 | 29 | 3 |
| Creatinine increased | 26 | 0 | 23 | 0 |
| Phosphate decreased | 21 | 3 | 13 | 2 |
| Calcium decreased | 21 | 0.6 | 29 | 0 |
LOW RATES OF CARDIAC DISORDERS, INCLUDING AFIB/FLUTTER, AND NO CARDIAC DEATHS AT THE ~3.5-YEAR MILESTONE vs IBRUTINIB*6
Afib/flutter
all grades
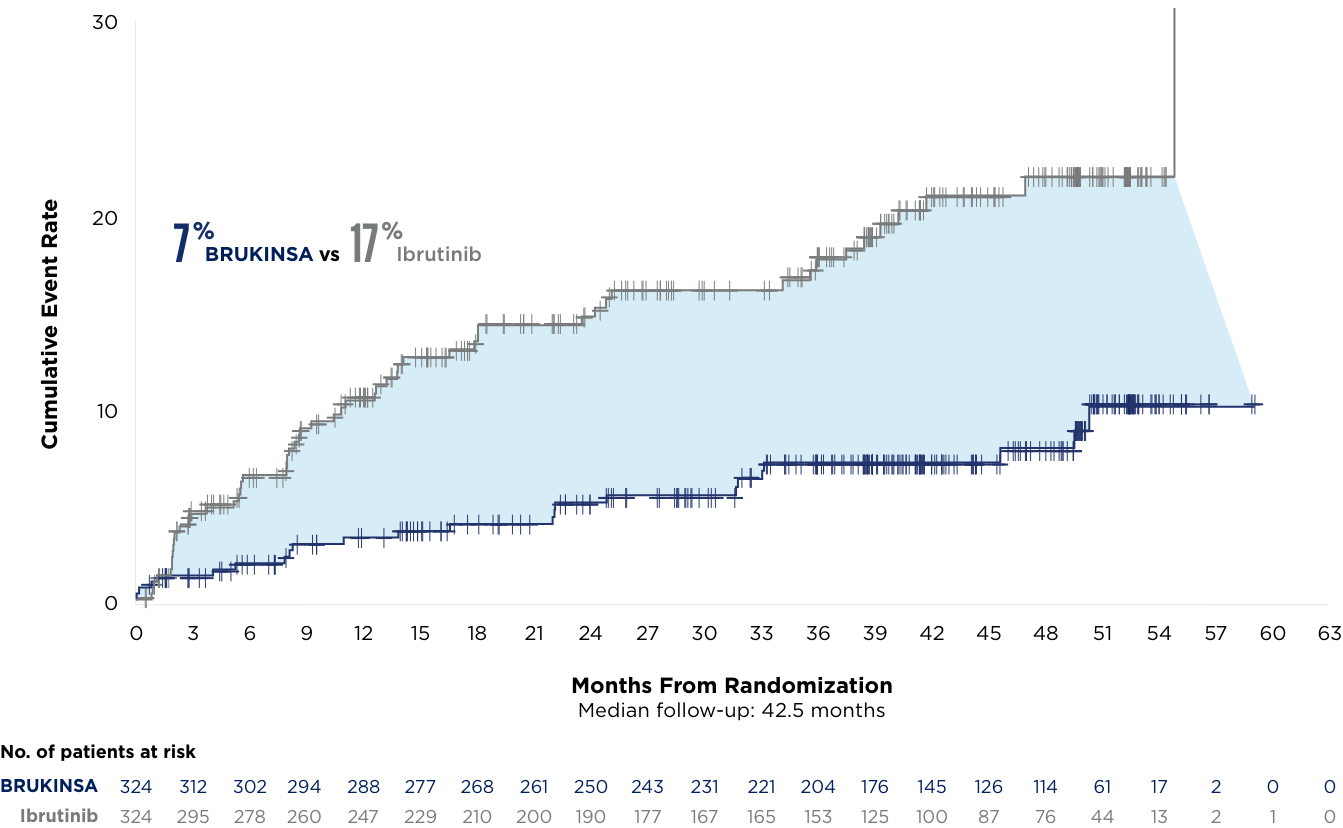
| Cardiac AEs | Initial Analysis5,7 | ~3.5-Year Milestone6 | ||
|---|---|---|---|---|
| BRUKINSA (n=324) | Ibrutinib (n=324) | BRUKINSA (N=324) | Ibrutinib (n=324) | |
| Cardiac AEs | 69 (21%) | 96 (30%) | 84 (26%) | 115 (36%) |
| Serious cardiac AEs | 6 (2%) | 25 (8%) | 13 (4%) | 32 (10%) |
| Cardiac AEs leading to treatment discontinuation | 1 (0.3%)* | 14 (4%)† | 3 (0.9%)‡ | 16 (5%)§ |
| Fatal cardiac events | 0 (0%) | 6 (2%) | 0 (0%) | 6 (2%) |
6 cardiovascular (CV) deaths occurred with ibrutinib in patients with and without a history of CV risk
No deaths occurred with BRUKINSA5,6
- Early onset death: 3 of the fatal cardiac events occurred within 4 months after initiation of ibrutinib in patients with coexisting cardiac conditions
- Late onset death: 3 of the fatal cardiac events occurred 2-3 years after the initiation of ibrutinib, including in 1 patient who had no history of cardiac disorders
SELECT ARs OF SPECIAL INTEREST IN ALPINE (STUDY 305)*1,2,5
| Adverse Reactions | ALPINE (Initial Analysis) | Pooled Safety Population† | ||||
|---|---|---|---|---|---|---|
| BRUKINSA (n=324) | Ibrutinib (n=324) | BRUKINSA (N=1729) | ||||
| All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | All Grades (%) | Grade ≥3 (%) | |
| Fatigue | 13 | 0.9 | 14 | 0.9 | 18 | 1 |
| Headache | 8 | 0 | 9 | 0 | 11 | 0.3 |
| Myalgia | 3 | 0 | 4 | 0 | 4 | 0.3 |
| Arthralgia | 14 | 0 | 15 | 0.3 | 14 | 0.6 |
| Atrial fibrillation and flutter | 5 | 3 | 13 | 4 | 4 | 2 |
| Hypertension | 19 | 13 | 20 | 13 | 14 | 7 |
| Major hemorrhage‡ | 4 | 3 | 4 | 4 | 5 | 4 |
ALPINE (STUDY 305): LOWER RATES OF DOSE REDUCTIONS AND DISCONTINUATION DUE TO ADVERSE EVENTS (AEs) AT THE INITIAL ANALYSIS5

Dr Anthony Nguyen discusses the safety profile of BRUKINSA vs ibrutinib in CLL
References: 1. BRUKINSA. Package insert. BeOne Medicines USA, Inc.; 2025. 2. Data on file. BeiGene USA, Inc. 3. Tam CS, Brown JR, Kahl BS, et al. Zanubrutinib versus bendamustine and rituximab in untreated chronic lymphocytic leukaemia and small lymphocytic lymphoma (SEQUOIA): a randomised, controlled, phase 3 trial. Lancet Oncol. 2022;23(8):1031-1043. 4. Tam CS, Munir T, Robak T, et al. Sustained efficacy of zanubrutinib vs bendamustine + rituximab in treatment-naive chronic lymphocytic leukemia/small lymphocytic lymphoma and continued favorable survival in non-randomized patients with del(17p): 6-year follow-up in the phase 3 SEQUOIA study. Presented at: American Society of Hematology (ASH) Annual Meeting and Exposition; December 6-9, 2025. Poster 2129. 5. Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib or ibrutinib in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2023;388(4):319-332. 6. Brown JR, Eichhorst B, Lamanna N, et al. Sustained benefit of zanubrutinib vs ibrutinib in patients with R/R CLL/SLL: final comparative analysis of ALPINE. Blood. 2024;144(26):2706-2717. 7. Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib demonstrates superior progression-free survival compared with ibrutinib for treatment of relapsed/refractory chronic lymphocytic leukemia and small lymphocytic lymphoma: results from final analysis of ALPINE randomized phase 3 study. Presented at: American Society of Hematology (ASH) Annual Meeting and Exposition; December 10-13, 2022.